Our Products
- REQUEST A -
QUOTE
- SCHEDULE A -
FREE DEMO
- ASK A -
QUESTION
About H&A Scientific, Inc.
For 30 years, H&A Scientific, Inc. has been a leader in complete laboratory software solutions. In 1992, our first software products, PC/Chrom and PC/Absorb, were DOS workstations for chromatography and atomic absorption, respectively. Since then, we have devoted our programming resources to the Microsoft Windows™ platforms.
In 1994, H&A released the ADIVP-1™ (Analog-to-Digital Interface Verification Package), a complete hardware/software product that verified the linearity, reproducibility, and accuracy of analog-to-digital (A/D) interfaces.
1996 saw the release of PC/Chrom for Windows™. PC/Chrom was a complete hardware/software product that interfaced your company’s GC or HPLC detector to a PC. That was followed by the release of PC/Chrom+ in 2001, a complete 32-bit rewrite of our earlier PC/Chrom for Windows™ and developed according to our industry-leading Quality Assurance Program.
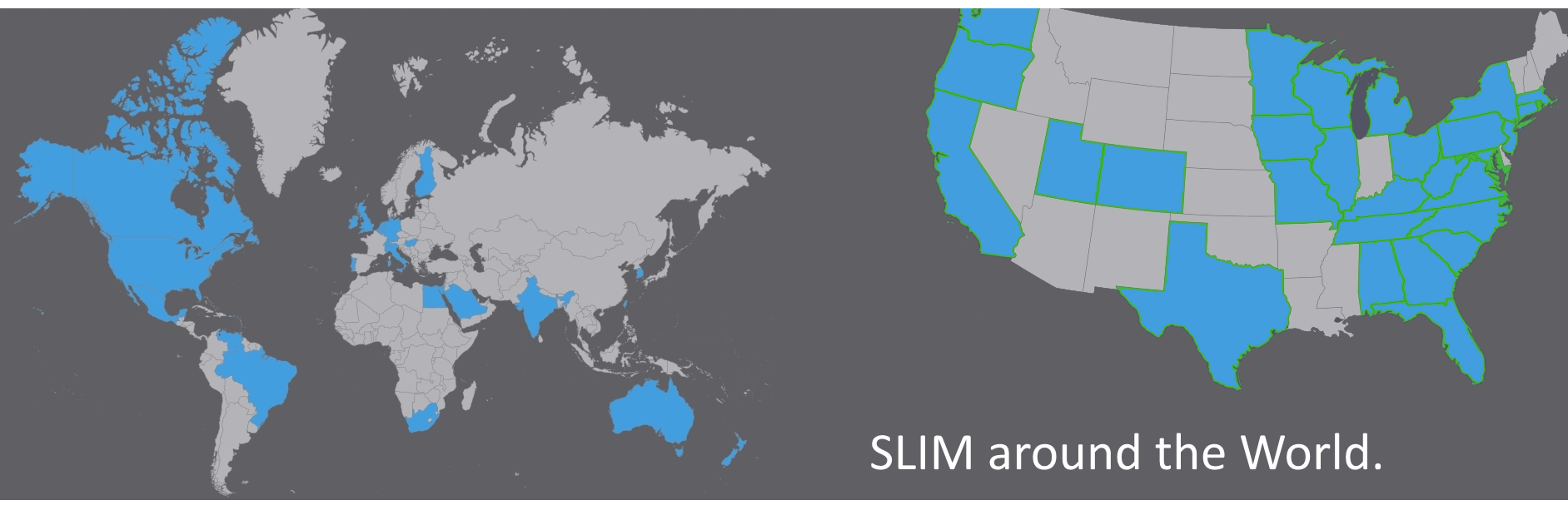
In 1997, H&A brought its SLIM (Stability Laboratory Information Manager) to market. A state-of-the-art software system, SLIM remains an industry leader stability study management software, from the design of stability study protocols to statistical analysis and presentation of data for final FDA submission. SLIM is a 32-bit client-server application. The client software operates on Windows, while the server software utilizes Microsoft SQL Server or Oracle.
H&A was busy in 1997 as it also launched SLIMStat, SLIM’s best-in-class statistical module, which was specifically designed to calculate a product’s shelf-life. Built with the same stability and ease-of-use H&A products are known for, it also provides trend analysis and accelerated condition calculations using the Arrhenius equation. Additionally, pooling of batches is supported per ICH Q1E. While SLIMStat is included as part of SLIM, it can also be sold and implemented as a stand-alone application.
As part of our continuing mission to serve the needs of stability professionals, H&A released the OPT-DISS, a High Yield Fiber Optic UV-Vis Dissolution System, in 1998. OPT-DISS is the result of a joint venture between H&A Scientific, Inc. and Distek.
2001 saw the debut of another class-leading product, IntelliFORM (Intelligent Fiber Optic Reaction Monitoring). IntelliFORM analyzed batch reactions to determine which reactions produced more product and had faster reaction rates. It used an innovative technique called Self-Modeling Curve Resolution to model reactions without the use of any products, allowing more information to be obtained at a lower cost.
In 2010, H&A Scientific was presented an export achievement certificate for accomplishments in the global marketplace by the United States Department of Commerce, U.S. Commercial Service. Our products have been sold and implemented in over 30 countries throughout the world, and are backed up by a support team of real experts and stability professionals — not call center personnel.